I remember the first time I heard the distinct, almost eerie silence of an electric car gliding past. It felt less like a vehicle and more like a whisper from the future. For years, the hum and rumble of gasoline engines defined our journeys, but now, a quiet revolution is underway, powered by a technology many of us take for granted: the electric vehicle (EV) battery. We hear about range, charging times, and environmental impact, but how does this marvel of modern engineering *actually* work? What secret alchemy transforms raw materials into the silent force propelling millions of cars around the globe?
It’s a question that’s fascinated me since I started seeing more and more charging stations pop up in my neighborhood. The switch from combustion to electricity isn't just about plugging in; it's a fundamental reimagining of personal transport, with the battery at its very heart. Understanding this core technology isn't just for engineers; it's for anyone curious about the future of mobility and our planet.
The Heart of the Matter: Lithium-Ion's Reign
At the core of almost every modern EV lies the **lithium-ion battery**. This isn't a new invention; it's been powering our laptops and smartphones for decades. But scaling it up for automotive use—demanding immense power, extended range, and rigorous safety—was a colossal engineering challenge.
So, what is a lithium-ion battery? In its simplest form, it consists of four main components:
- Cathode: The positive electrode, typically made from a lithium metal oxide (e.g., lithium nickel manganese cobalt oxide - NMC, or lithium iron phosphate - LFP). This is where lithium ions are stored when the battery is discharged.
- Anode: The negative electrode, usually made of graphite. This stores lithium ions when the battery is charged.
- Electrolyte: A liquid or gel material that allows lithium ions to move between the anode and cathode. It’s the highway for the ions.
- Separator: A thin, porous membrane that physically separates the anode and cathode to prevent short circuits while still allowing ions to pass through the electrolyte.
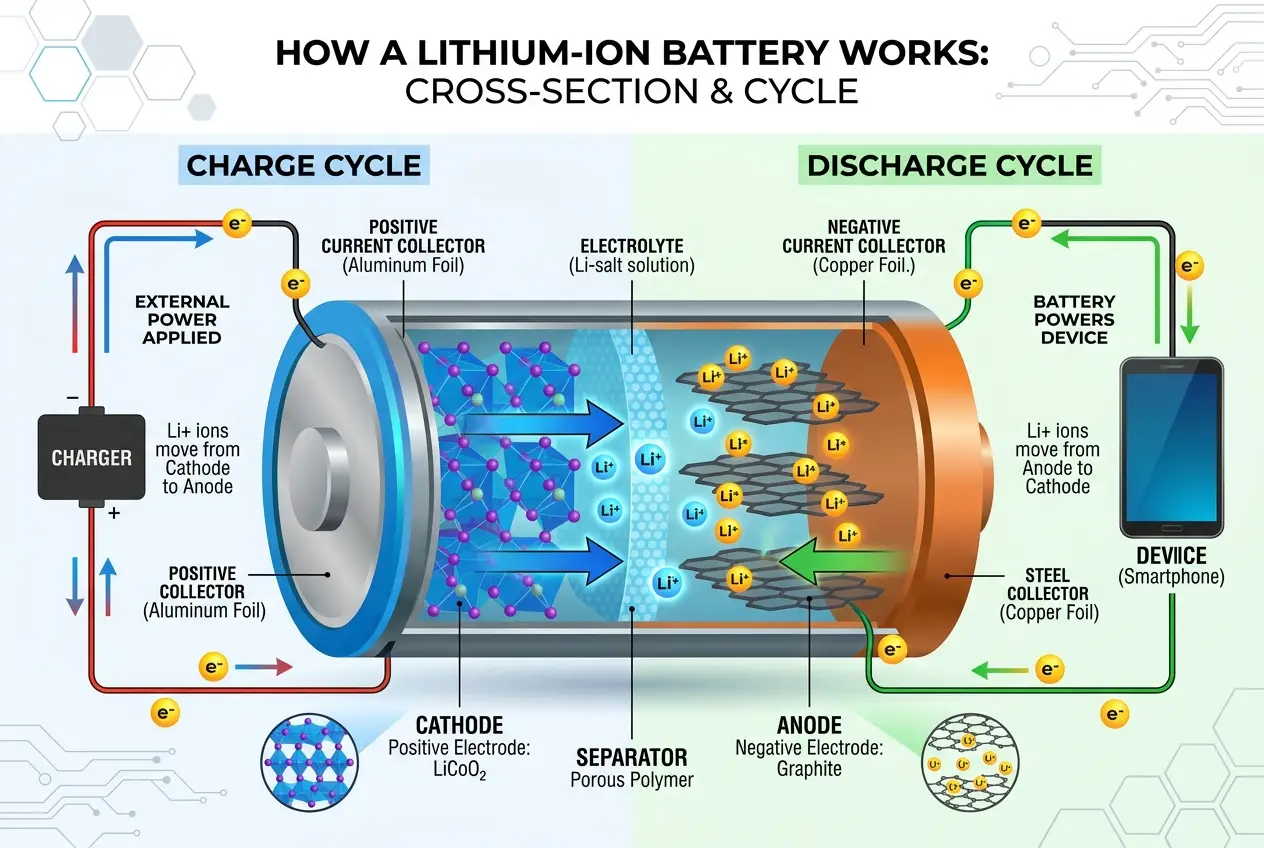
When you charge an EV, lithium ions move from the cathode through the electrolyte and separator to the anode, where they are stored. This process requires energy. When you drive, the reverse happens: the ions flow back from the anode to the cathode, releasing electrons which then flow through an external circuit (your car's motor), creating the electric current that powers the vehicle. This flow of electrons is what we call electricity. For a deeper dive into the electrochemical processes, you can refer to the detailed explanations on
Wikipedia's Lithium-ion battery page.
From Cell to Pack: Building Automotive Powerhouses
A single lithium-ion cell, like those in your phone, wouldn't be enough to power a car. Instead, thousands of these cells are grouped together to form a robust **battery pack**. This assembly happens in stages:
1. **Cells:** Individual battery units, typically cylindrical (like AA batteries, but larger), pouch (flat, rectangular), or prismatic (hard-cased rectangular).
2. **Modules:** Multiple cells are wired together into a module, often encased for protection and thermal management. These modules might contain dozens of cells.
3. **Pack:** Several modules are then combined into a large battery pack, which is the complete unit installed in the car's chassis. The pack also includes vital control systems, cooling systems, and safety features.
The sheer scale of an EV battery pack is impressive. For instance, a Tesla Model S might use thousands of individual cylindrical cells, while other manufacturers opt for larger pouch or prismatic cells.
The Unsung Hero: The Battery Management System (BMS)
The battery pack isn't just a collection of cells; it's an intelligent system. The **Battery Management System (BMS)** is the brain of the operation. I can't stress enough how critical the BMS is for the longevity, safety, and performance of an EV battery. Its functions include:
* **Monitoring:** Constantly tracking individual cell voltage, temperature, and current flow.
* **Balancing:** Ensuring all cells charge and discharge uniformly to prevent overcharging or undercharging, which can degrade the battery or pose safety risks.
* **Protection:** Guarding against overcurrent, overvoltage, undervoltage, and overheating.
* **Thermal Management:** Actively cooling or heating the battery to keep it within its optimal operating temperature range, which is crucial for efficiency and lifespan.
* **State of Charge (SoC) and State of Health (SoH) Calculation:** Providing accurate estimates of remaining charge and overall battery degradation.
Without a sophisticated BMS, the powerful energy stored in an EV battery would be unstable and short-lived. This intelligent control is a testament to modern electronics and software engineering.
Charging Up: More Than Just Plugging In
When you plug in an EV, you're initiating a complex dance between the charging infrastructure, the car's onboard charger, and the BMS. There are two main types of charging:
* **AC Charging (Level 1 & 2):** This is common for home charging. The alternating current (AC) from the grid is converted to direct current (DC) by the car's **onboard charger**. Level 1 uses a standard household outlet (slow), while Level 2 uses a higher-voltage outlet or dedicated charging station (faster).
* **DC Fast Charging (Level 3):** These are public rapid chargers. Here, the AC-to-DC conversion happens *outside* the car, in the charging station itself. This allows for much higher power delivery directly to the battery, significantly reducing charging times. For more information on charging standards and technologies,
Wikipedia's Charging station page offers a comprehensive overview.
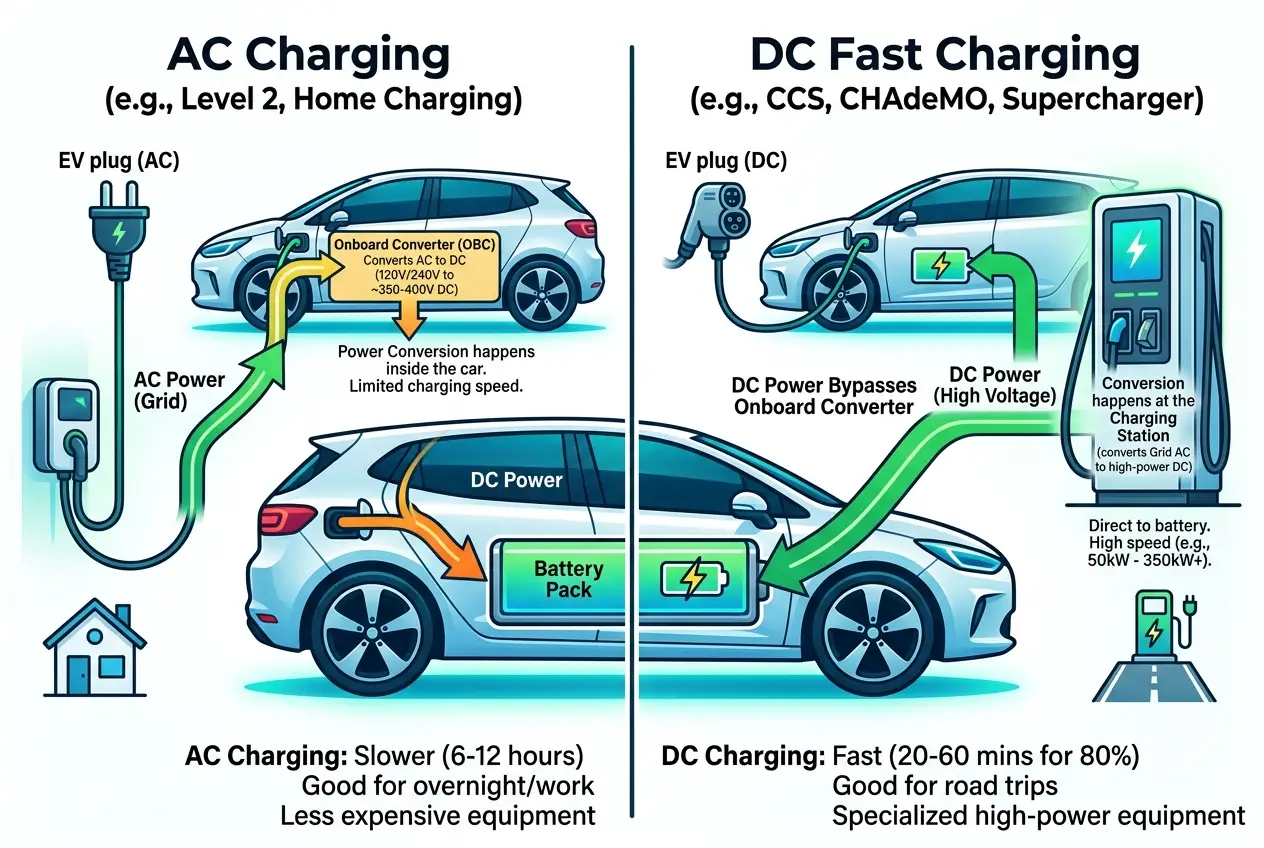
The speed of charging also depends on the battery's voltage architecture (e.g., 400V or 800V) and its ability to accept power without overheating or damaging the cells. Modern EVs with 800V systems can achieve incredibly fast charging speeds, adding hundreds of miles of range in minutes.
Decoding Range Anxiety: What Affects How Far You Go?
"Range anxiety" is a common concern for potential EV owners, but understanding the factors that influence an EV's range can help alleviate it.
* **Battery Capacity (kWh):** This is the most straightforward factor. A larger battery (higher kilowatt-hour rating) stores more energy and generally offers more range. Think of it like the size of your car's fuel tank.
* **Vehicle Efficiency:** Just like gasoline cars, EVs have varying efficiencies. Aerodynamics, vehicle weight, tire choice, and the efficiency of the electric motors all play a role.
* **Temperature:** Both extremely cold and hot temperatures can impact range. Cold weather reduces battery performance and requires energy for cabin heating, while hot weather can stress the battery and require more energy for cooling.
* **Driving Style:** Aggressive driving, high speeds, and frequent hard acceleration consume more energy and reduce range, whereas smooth, consistent driving and regenerative braking can extend it.
The Road Ahead: Beyond Lithium-Ion
While lithium-ion batteries are excellent, research is constantly pushing the boundaries. I'm personally very excited about the advancements that could reshape our electric future. Some promising technologies include:
Solid-State Batteries
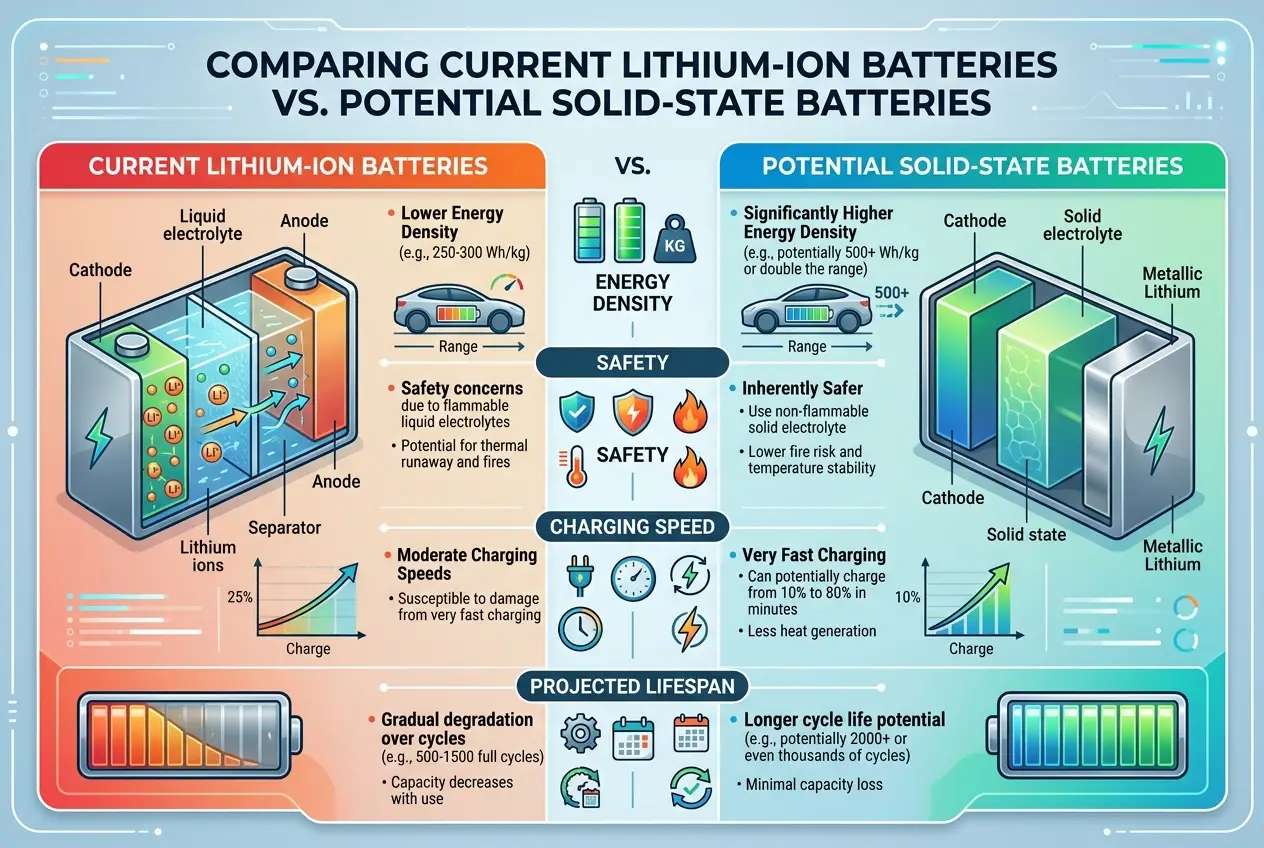
This is often touted as the "holy grail" of battery technology. Instead of a liquid electrolyte, solid-state batteries use a solid material. This promises:
* **Higher Energy Density:** More range for the same size/weight.
* **Improved Safety:** Eliminates the flammable liquid electrolyte, reducing fire risk.
* **Faster Charging:** Potentially much quicker charging times.
* **Longer Lifespan:** Better cycle stability.
However, challenges remain in manufacturing these batteries at scale and ensuring reliable performance over wide temperature ranges. For a closer look at potential future battery technologies, you might find our previous blog on
Can Diamond Batteries Power Our Future? intriguing.
Other Promising Chemistries and Innovations
* **Sodium-Ion Batteries:** Sodium is far more abundant and cheaper than lithium. While currently offering lower energy density, they could be excellent for less demanding applications or as a cheaper alternative for entry-level EVs.
* **Silicon Anodes:** Replacing graphite with silicon in anodes could significantly increase energy density. Silicon can store much more lithium, but it expands and contracts, posing engineering challenges.
* **Advanced Cathode Materials:** Ongoing research into new cathode materials aims to improve performance, reduce reliance on rare or controversial materials, and enhance safety. This often involves exploring how
exotic materials can reshape electronics.
* **Battery Recycling and Second Life:** As EVs become more widespread, managing used batteries is crucial. Recycling processes are improving to recover valuable materials. Additionally, retired EV batteries can be repurposed for grid storage or other less demanding applications, extending their useful life.
Conclusion: The Quiet Revolution Continues
The electric vehicle battery is far more than just a power source; it's a testament to human ingenuity and a cornerstone of our sustainable future. From the intricate electrochemical dance within each cell to the sophisticated management systems that oversee an entire pack, these batteries are driving a profound shift in how we move, how we generate, and how we consume energy.
As the technology continues to evolve, promising even greater range, faster charging, and enhanced sustainability, I believe we're only just beginning to tap into the full potential of electric mobility. The quiet revolution that began with a silent car passing on the street is now reshaping industries and daily lives, propelling us towards a cleaner, more efficient tomorrow. It makes me wonder what other technological marvels, like those that could potentially help
AI build our first city on the moon, are just around the corner, waiting for their own power source to be perfected.

 Join Us
Join Us






































Leave a Reply
Comments (0)